SUNSTAR Periocline®
Description:
Sunstar Periocline 2%, Dental Ointment, contains a local therapeutic antibiotic agent which acts in the treatment of moderate to severe chronic adult periodontal disease. The active ingredient minocycline hydrochloride, is gradually released from the microsphere formulation in-situ after injection of the formulation into the gingival sulcus. Minocycline has a high affinity for the surface of the tooth and therefore remains in situ after release. Therefore, it provides a simple-to-apply, sustained release adjunctive treatment for periodontitis.
Available in Singapore, Thailand, Indonesia
Share with your friends:
- Easy to Handle localized antibiotic agent
- Sustained Release Drug Delivery System
- Once every 2-weeks administration
- Excellent Antimicrobial agent
- Excellent Clinical Results
Active ingredient: minocycline
Periocline contains the active ingredient minocycline at a concentration of 2%. Minocycline is one of a group of antibiotics called tetracyclines.
LOCALIZED ACTION
- With targeted antimicrobial efficiency on (1,2):
- Anaerobes: Porphyromonas gingivalis, Prevotella intermedia, Tannerella forsythia, Eikenella corrodens, Aggregatibacter actinomycetemcomitans
- Spirochetes: Treponema denticola
- High concentrations in-situ, low systemic concentrations (3)
- In a pharmacokinetic study, concentrations of minocycline in gingival crevicular fluid remained at clinical-effective levels for a minimum of three days following the administration of 0.05 g of gel (1 mg minocycline) into periodontal pockets.
- Serum levels after subgingival administration and even after direct oral administration of 0.5 g of gel (10 mg minocycline) were only in the range of 0.1-0.2 µg/ml. These levels are much lower than the serum levels which would be associated with the normal oral dose of 100-200 mg/day minocycline used in systemic therapy.
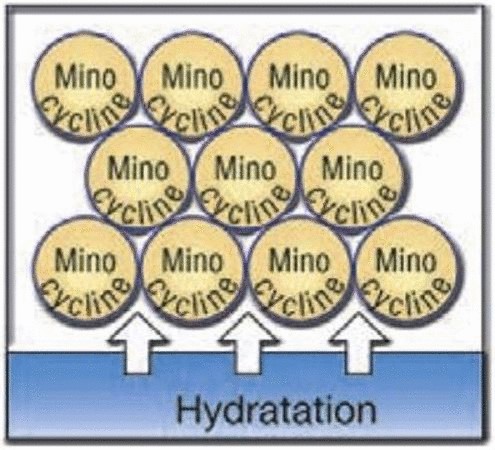
- Sustained release drug-delivery systemPeriocline is a light yellow-coloured ointment. The exclusive process of micro encapsulation allows continuous, regular and slow release of the minocycline drug over several days. Active ingredient minocycline hydrochloride is gradually released in situ from the microsphere formulation after application of the gel into the periodontal pocket. Moreover, minocycline has a high affinity for the surfaces of the tooth and therefore remains available in situ after release from the formulation. Therefore, GUIDOR Periocline provides a simple-to-apply, sustained release adjunctive treatment for periodontitis.
References:
- Satomi et al. Minocycline HCl concentration in periodontal pocket after administration of LS007. Journal of Japanese Society of Periodontology, 1987; 29(3) : 937-43
- Mc Coll E, Patel K, Dahlen G, Tonetti M, Graziani F, Suvan J, Laurell L. Supportive periodontal therapy using mechanical instrumentation or 2 % minocycline gel : a 12 month randomized controlled, single masked pilot study. J Clin Periodontol. 2006 ; 33 : 141-50
- SmPC